The urgent need to identify viable treatments and a potential vaccine for SARS-CoV-2 infection has underscored the importance of translating insights from basic science into clinical application.
Multiple recent studies have effectively utilized organoids as pre-clinical models for new infectious diseases, including a widely praised study authored by an international group of researchers led by LSI Director Josef Penninger.
This work was highlighted in a recent overview published in Nature Reviews Molecular Cell Biology. “To understand the tissue tropism of SARS-CoV-2, multiple research groups resorted to organoid approaches,” observed author Hans Clevers, a Dutch molecular biologist. “Penninger and colleagues demonstrated that SARS-CoV-2 could directly infect capillary organoids and kidney organoids, both established from human iPSCs. These observations may explain the spread of the virus through the body and the loss of kidney function in severely ill individuals.”
“I have been familiar with organoids for many years, and I was involved in the development of the first human brain organoid,” Dr. Penninger said in a separate interview. “More recently, Reiner Wimmar from my group developed blood vessel organoids from a single stem cell which was published in Nature last year.
“For the recent COVID-19 findings, we had help with the kidney organoids from Núria Montserrat, who is an organoid expert in Barcelona, and Ali Mirazimi, who studies infectious diseases in Stockholm. And of course the intestine was an area of interest, because we know from our previous findings that ACE2 affects intestinal uptake of amino acids by regulating the renin-angiotensin pathway. This is where Ryan Conder’s expertise with intestinal organoids became really helpful, as he worked with us and helped generate these organoids for our study. We continue to work on these organoids and very interesting data will be published soon. Together with these teams we published our recent Cell paper in about five weeks, an unusually short amount of time.”
STEMCELL’s role is in these studies has been very important, according to Dr. Penninger, “because we need to study the pathology of the virus, and this is not fully possible in patients. Organoids are the closest we can get to real human tissues. We can study how the virus interacts with more complex organoids that mimic organs. Studying the disease in mouse models can also be helpful, but testing therapeutics in mice usually cannot replace testing in human tissue. We can even potentially test vaccines in these systems, which still requires more work, to see if we can prevent infections in different organs.
“I hope that we can work together even more closely to strengthen our academic-industrial collaboration,” Dr. Penninger added. “I think there is a lot that could come from academia, and I think you guys are amazing people who make intestinal and many other organoids, and STEMCELL can contribute a lot.”
“Collaborating to Accelerate COVID-19 Research” produced by STEMCELL Technologies, highlights the contributions of Dr. Penninger and the international team behind “Inhibition of SARS-CoV-2 Infections in Engineered Human Tissues Using Clinical-Grade Soluble Human ACE2.” Dr. Penninger begins speaking at 1:48.
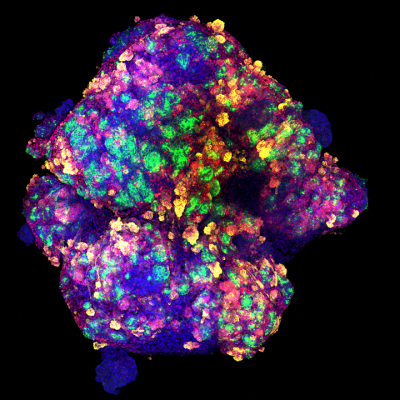
Cover image of a kidney organoid courtesy of Núria Montserrat
Portions of this story are drawn from a profile of Dr. Penninger produced by STEMCELL Technologies