An international team led by LSI Director Dr. Josef Penninger has found a trial drug that effectively blocks the cellular door SARS-CoV-2 uses to infect its hosts.
The findings, published today in Cell, hold promise as a treatment capable of stopping early infection of the novel coronavirus that, as of April 2, has affected more than 981,000 people and claimed the lives of 50,000 people worldwide.
The study provides new insights into key aspects of SARS-CoV-2, the virus that causes COVID-19, and its interactions on a cellular level, as well as how the virus can infect blood vessels and kidneys.
“We are hopeful our results have implications for the development of a novel drug for the treatment of this unprecedented pandemic,” says Penninger, professor in UBC’s faculty of medicine, and the Canada 150 Research Chair in Functional Genetics at UBC.
“This work stems from an amazing collaboration among academic researchers and companies, including Dr. Ryan Conder’s Gastrointestinal Group at STEMCELL Technologies in Vancouver, Nuria Montserrat in Spain, Drs. Haibo Zhang and Art Slutsky from Toronto and especially Ali Mirazimi’s infectious biology team in Sweden, who have been working tirelessly day and night for weeks to better understand the pathology of this disease and to provide breakthrough therapeutic options.”
ACE2 — a protein on the surface of the cell membrane — is now at centre-stage in this outbreak as the key receptor for the spike glycoprotein of SARS-CoV-2. In earlier work, Penninger and colleagues at the University of Toronto and the Institute of Molecular Biology in Vienna first identified ACE2, and found that in living organisms, ACE2 is the key receptor for SARS, the viral respiratory illness recognized as a global threat in 2003. His laboratory also went on to link the protein to both cardiovascular disease and lung failure.
While the COVID-19 outbreak continues to spread around the globe, the absence of a clinically proven antiviral therapy or a treatment specifically targeting the critical SARS-CoV-2 receptor ACE2 on a molecular level has meant an empty arsenal for health care providers struggling to treat severe cases of COVID-19.
“Our new study provides very much needed direct evidence that a drug — called APN01 (human recombinant soluble angiotensin-converting enzyme 2 – hrsACE2) — soon to be tested in clinical trials by the European biotech company Apeiron Biologics, is useful as an antiviral therapy for COVID-19,” says Dr. Art Slutsky, a scientist at the Keenan Research Centre for Biomedical Science of St. Michael’s Hospital and professor at the University of Toronto who is a collaborator on the study.
APEIRON Biologics today announced that it has received regulatory approvals in Austria, Germany and Denmark to initiate a Phase II clinical trial of hrsACE2, (known as APN01) to treat COVID-19.
In cell cultures analyzed in the current study, hrsACE2 inhibited the coronavirus load by a factor of 1,000-5,000. In engineered replicas of human blood vessel and kidneys — organoids grown from human stem cells — the researchers demonstrated that the virus can directly infect and duplicate itself in these tissues. This provides important information on the development of the disease and the fact that severe cases of COVID-19 present with multi-organ failure and evidence of cardiovascular damage. Clinical grade hrsACE2 also reduced the SARS-CoV-2 infection in these engineered human tissues.
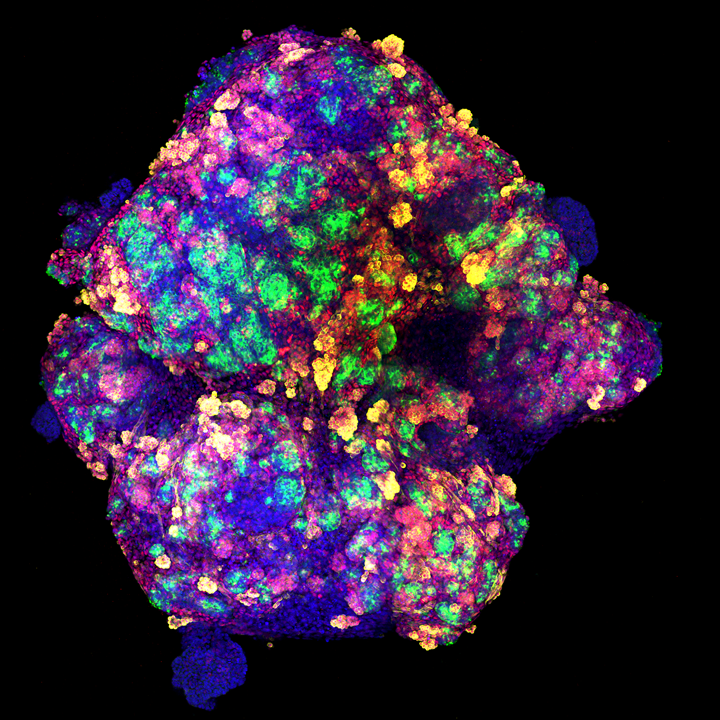
Representative image of a kidney organoid generated in vitro using human pluripotent stem cells. Photo Credit: Nuria Monserrat*
“Using organoids allows us to test in a very agile way treatments that are already being used for other diseases, or that are close to being validated. In these moments in which time is short, human organoids save the time that we would spend to test a new drug in the human setting,” says Núria Montserrat, ICREA professor at the Institute for Bioengineering of Catalonia (IBEC) in Spain.
“The virus causing the COVID-19 is a close sibling to the first SARS virus,” adds Penninger. “Our previous work has helped to rapidly identify ACE2 as the entry gate for SARS-CoV-2, which explains a lot about the disease. Now we know that a soluble form of ACE2 that catches the virus away, could be indeed a very rational therapy that specifically targets the gate the virus must take to infect us. There is hope for this horrible pandemic.”
This research was supported in part by the Canadian federal government through emergency funding focused on accelerating the development, testing, and implementation of measures to deal with the COVID-19 outbreak.
About LSI
The Life Sciences Institute (LSI) at the University of British Columbia conducts basic research aimed at improving human and planetary health. Home to 17 prestigious Canada Research Chairs, the LSI hosts multidisciplinary teams working to develop a comprehensive understanding of living organisms and systems. Members of our nine research groups conduct world-class research in diabetes, cardiovascular disease, immune response, infectious disease, cancer biology, developmental disorders, bacterial regulation, neuroscience, structural biology, environmental microbiology and molecular epigenetics.
Launched in 2005 in association with the UBC Faculties of Science and Medicine, LSI houses the vibrant research labs of 87 investigators drawn from 15 departments across five faculties. We collaborate in the 270,000 s.f. Life Sciences Centre, where seven state-of-the-art core facilities – ranging from flow cytometry to cryo-electron microscopy and bioformatics – support discoveries that hold promise of direct health benefits for Canadians and Canada.
About UBC
The University of British Columbia is a global centre for research and teaching, consistently ranked among the top 20 public universities in the world. Since 1915, UBC’s entrepreneurial spirit has embraced innovation and challenged the status quo. UBC encourages its students, staff and faculty to challenge convention, lead discovery and explore new ways of learning. At UBC, bold thinking is given a place to develop into ideas that can change the world.
About St. Michael’s
St. Michael’s Hospital provides compassionate care to all who enter its doors. The hospital also provides outstanding medical education to future health care professionals in more than 27 academic disciplines. Critical care and trauma, heart disease, neurosurgery, diabetes, cancer care, care of the homeless and global health are among the Hospital’s recognized areas of expertise. Through the Keenan Research Centre and the Li Ka Shing International Healthcare Education Centre, which make up the Li Ka Shing Knowledge Institute, research and education at St. Michael’s Hospital are recognized and make an impact around the world. Founded in 1892, the hospital is fully affiliated with the University of Toronto.
About STEMCELL Technologies
STEMCELL Technologies is Canada’s largest biotechnology company, with over 1,500 employees and year-on-year growth of approximately 20% for the last 26 years. Based in Vancouver, STEMCELL supports life sciences research around the world with more than 2,500 specialized reagents, tools, and services. STEMCELL offers high-quality cell culture media, cell separation technologies, instruments, accessory products, and educational resources that are used by scientists advancing the stem cell, immunology, cancer, regenerative medicine, microbiology and cellular therapy fields. Find more information at www.stemcell.com.
About the Institute for Bioengineering of Catalonia
The Institute for Bioengineering of Catalonia (IBEC) is a leading-edge multidisciplinary research centre based in Barcelona that conducts research at the frontiers of basic and life sciences linked with engineering to generate new knowledge and applications that helps to enhance health and quality of life. IBEC create wealth by putting together biophysics, cell engineering, nanomedicine, biomaterials, tissue engineering and the applications of information technology to health. IBEC is a non-profit-making foundation set up in 2005 by the Departments of Health and Innovation, Universities and Enterprise of the Government of Catalonia, the University of Barcelona and the Technical University of Catalonia. http://www.ibecbarcelona.eu/
*Representative image of a kidney organoid generated in vitro using human pluripotent stem cells. The procedure takes advantage of 3D culture using specific renal inductive signals, a methodology that only lasts 20 days. Proximal tubule-like structures are detected by LTL staining (in green) and podocyte-like cells are characterized by the co-expression of WT1 (in red) and podocalyxin [PODXL] (in yellow). Nuclei are detectedby DAPI staining (in blue).
Photo credit: Nuria Monserrat ICREA professor at the Institute for Bioengineering of Catalonia (IBEC) in Spain.
Photo credit main image: Tibor Kulcsar, copyright imp-IMBA graphics department 2020